EU MDR Consulting for
Japanese Medical Device Manufacturers
Belgium-based regulatory support for CER preparation, Authorised Representative
arrangements, Legal Representative & CRO management, and European market compliance.
Our Services
End-to-end EU MDR compliance and European market access support
CER & Clinical Evaluation
Systematic literature search (PubMed, EMBASE), SOTA analysis, equivalent device assessment, and CER drafting in line with MDCG 2020-1 and 2020-13.
Authorised Representative Setup
3New SRL is registered as an AR in EUDAMED. We support AR selection, evaluation, contract structuring, and ongoing AR management under Article 11 MDR. AR mandate subject to individual review.
Legal Representative & CRO Management
For manufacturers conducting Clinical Investigations in Europe: Legal Representative engagement, CRO selection and oversight, NDA/IP review, and contract structuring with European partners.
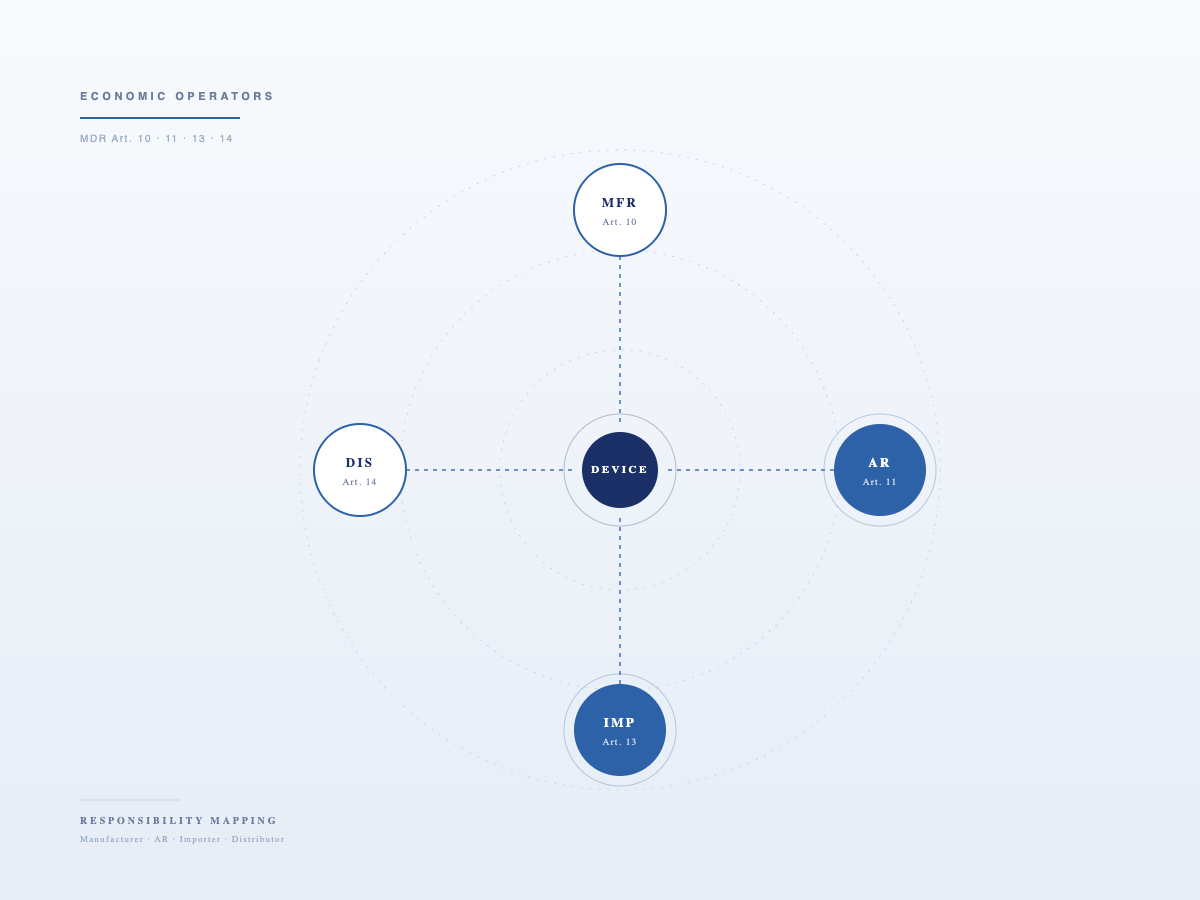
Economic Operator Audit Support
MDR compliance gap analysis for AR, Importer, and Distributor against Articles 11, 13, and 14. Audit preparation, checklist development, and corrective action support.
GDPR & European Market Setup
GDPR gap identification for clinical and PMS activities, Data Processing Agreement review, and guidance on establishing a Belgian or European entity.
Why 3New SRL
Based in Belgium, at the heart of the EU
We are incorporated in Belgium with direct working relationships with Notified Bodies, Competent Authorities, CROs, European investigators, and distributors. On-the-ground knowledge informs every recommendation.
Bridge between Japan and Europe
We work with Japanese regulatory affairs, quality assurance, and international business teams. Our principal communicates in Japanese and understands both regulatory cultures — eliminating the need for a separate interpreter layer.
Practical, usable deliverables
We focus on templates, checklists, and procedures your team can put to work immediately — not generic advice. MDCG guidance is translated into actionable steps.
IP-aware contract support
Our principal holds a Japanese patent attorney qualification. We apply this not to IP prosecution, but to reviewing confidentiality, data ownership, and deliverable rights in CRO, investigator, and distributor agreements.
Authorised Representative
3New SRL is registered as an Authorised Representative in EUDAMED.
AR mandates are assessed case by case, based on device type, risk classification, technical documentation status, existing CE certification, insurance, PRRC/QMS setup, and the scope of the manufacturer's mandate.
Our current AR-related services include:
- AR candidate selection and evaluation
- Existing AR arrangement gap analysis
- AR contract and role clarification
- AR SOP and QMS documentation support
Key AR Responsibilities under Article 11 MDR
- Maintaining and making available the EU Declaration of Conformity, technical documentation, and certificates to Competent Authorities
- Verifying the manufacturer's EUDAMED registration and UDI obligations
- Relaying complaints and vigilance information between the manufacturer and authorities
- Acting as the manufacturer's contact point with Competent Authorities
- Taking appropriate action if the manufacturer fails to fulfil MDR obligations
Primary obligations for product registration and vigilance reporting rest with the manufacturer. The AR confirms, facilitates, and supports authority-facing activities.
About 3New SRL

3New SRL is a Belgium-incorporated regulatory consulting company dedicated to supporting Japanese medical device manufacturers in EU MDR compliance, clinical evaluation, and European market access. We combine on-the-ground presence in Belgium with the ability to work closely with Japanese industry teams.
| Company | 3New SRL |
|---|---|
| Location | Belgium (European Union) |
| Services | EU MDR compliance, CER preparation, AR setup, Legal Representative & CRO management, Economic Operator audit, GDPR, European market access |
| Primary clients | Japanese medical device manufacturers — RA, QA, and international business teams |
| Principal qualification | Japanese patent attorney (applied to contract and IP risk management, not IP prosecution) |
We do not offer legal advice, medical opinions, or representation before regulatory authorities. For legal matters, please engage a qualified lawyer.
Contact Us
An initial overview discussion is available free of charge. Tell us about your situation and we'll explain how we can help.
Typical enquiries we receive
- Starting EU MDR compliance — where to begin
- CER preparation or literature search outsourcing
- Authorised Representative selection, evaluation, or contract review
- Legal Representative and CRO management for clinical investigations
- MDR compliance check for European distributors or importers
- GDPR gap assessment for clinical or PMS data activities
- Belgium or European entity setup
Detailed document review, regulatory assessment, contract review, and analysis are provided as paid services.
Send us an emailRepresentative Profile
View on LinkedInDisclaimer
The information on this website is provided for general informational purposes only and does not constitute legal advice, medical advice, or regulatory representation, and does not guarantee CE marking approval or Notified Body audit success.
An initial overview discussion is available free of charge. Detailed document review, regulatory assessment, contract review, investigation, or analysis are provided as paid services. Regulatory information (EU MDR, MDCG guidance) is subject to change; always verify against current primary sources.
3New SRL's scope does not include legal advice from a lawyer or law firm, medical judgement from a physician, or certification by a Notified Body or Competent Authority.